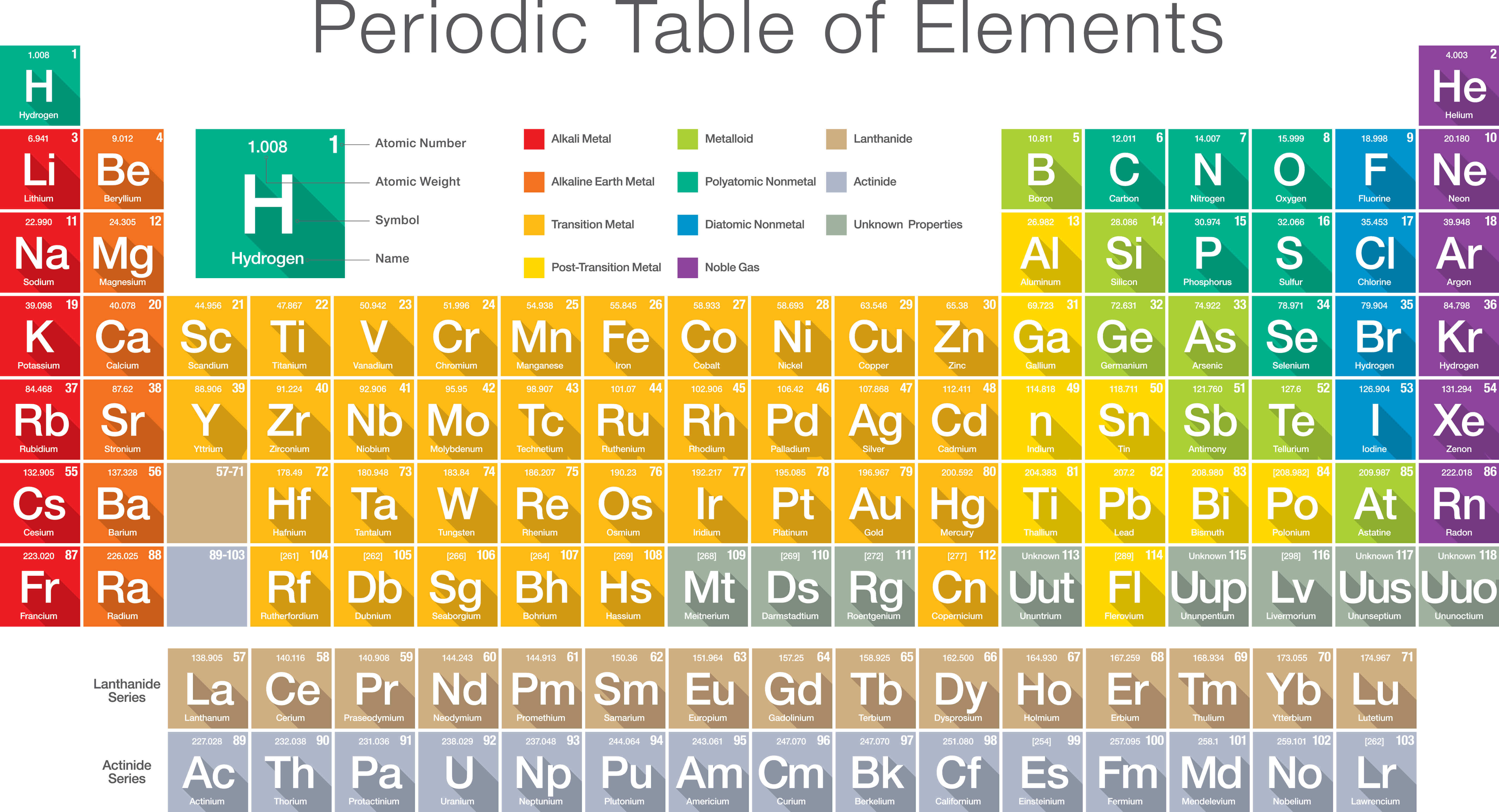
 Reaction of Non-metals with Hydrogen: When non-metals react with hydrogen, they form covalent hydrides.Non-metal + Chlorine → Non-metal chloride Reaction of Non-metal with Chlorine: Non-metals react with chlorine gas to produce chlroide.Reaction of Non-metals with Oxygen: Non-metals form oxide when reacting with oxygen.Colour: Non-metals are present in many colours.Density: Most of the non-metals have low density.Melting and boiling point: Non-metals generally have low melting and boiling points except Diamond which has a very high melting and boiling point.Malleability and ductility: Non-metals are brittle.Graphite, which is an allotrope of Carbon, is an exception to this as it is a good conductor of electricity. Conduction: Non-metals are a bad conductor of electricity and heat.Lustre: All non-metals have a dull appearance except Diamond and iodine.State: Non-metals may be solid, liquid, or gas.However, diamond is an exception to this as it is the hardest substance that occurs naturally. Softness: Non-metals are generally soft.Thus, non-metals are also known as Electronegative Elements. Non-metals form negative ions by gaining an electron. On tapping with a hammer, non-metals break down into very fine thin powdery mass.įew of the non-metals are Carbon, Oxygen, Sulphur, Nitrogen (N), Chlorine (Cl), Bromine (Br), Neon (Ne), and Argon (Ar), etc. /GettyImages-1154261034-08fa91cb3d8942c093b9e6b66a26f690.jpg)
Non-metals are very soft and dull in appearance. Non-metals are neither malleable nor ductile. Non–metals do not exhibit the characteristics of metals, i.e.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
